 
The note of caution in your ( ) first sentence is then substantially laid to rest: With that established: The simple 2-energy level system does have the usual relationship where increasing its energy remains synonymous with increasing its temperature. Where the gradient = 1/T is monotonically decreasing with E. The temperature is negative in that region but perfectly constant.įortunately the entropy of the simple 2-energy level system doesn't look like that, it has this sort of shape:Įntropy2.png (24.19 kB. 1152圆48 - viewed 1989 times)Īn increase in the the total energy of the system would not produce an increase in temperature once you've entered the region with most of the particles in the higher energy state. Suppose the entropy, S, of a system of N particles (each particle having only two energy levels) varied with E, the total energy of the system, as follows: 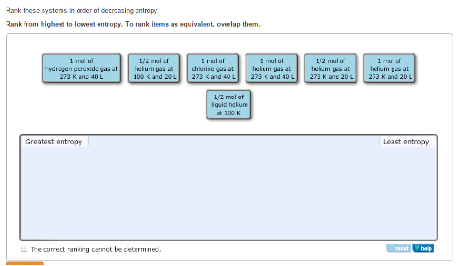
For a more arbitrary substance and where statistical temperature is used, you really do have to directly check the gradient ∂S/∂E. As you ( ) implied it's clear that energy is always being added to the system as more particles are driven to the higher energy state, however that wouldn't guarantee that the temperature was increasing.įor a typical substance like an ideal gas held at constant volume, it's apparent that increasing the internal energy will increase the temperature. The situation with a simple two-energy level system does illustrate a situation where temperature can increase (in this case it is increasing by becoming less negative) while the entropy decreases. As you ( ) implied: It's very much a case of needing to keep our definitions of temperature straight (and it does become a little dull then). if the energy is increasing, and the entropy decreasing, then the system has a negative temperature (ie below absolute zero) One reasonable definition of Temperature is: T = ∂U/∂S (constant volume). Think this isn't realistic? It's how lasers work.Īlso, going back to my initial remark about temperature: I never said anything about the temperature of the system of bits, only that you are adding energy. But once the 50/50 state is reached, any additional energy going in is going to decrease the entropy of the system, until finally it is 100% energized, and has regained its initial low entropy. Initially the entropy increases with increasing temperature. Imagine all the bits start out in the low energy state, and you start feeding energy into the system. More broadly, we can imagine any system made up of bits that can be in two states (one high energy, one low). If some light is absorbed, the resulting system has both more energy and less entropy than it did before (it's not a closed system). The first example that springs to my mind is the electronic structure of a transition metal ion. There are plenty of systems one can imagine (or even observe!) in which increasing energy of the system results in there being fewer available micro-states, and therefore lower entropy. I think you need to be careful about specifying whether you mean temperature or thermal energy. 
Also, using a statistical mechanics definition of entropy you don't seem to be asking for too much: If there was a substance where you can increase the total energy content from E i to E f but the number of microstates availble for total energy E f is less than the number of microstates available for total energy E i - then you would seem to be on to something.) I'm asking because I don't know for certain if there is a substance with C v < 0. (It doesn't seem possible to me, using a thermodynamic definition of entropy, just because C v = specific heat capacity at constant volume is always positive for every substance I know about. 
Now if we insist that all changes are restricted to maintaining a constant volume, we have the question that I was really hoping to ask: Is there a substance where the temperature can be increased but the entropy of that substance decreases and all changes are done at constant volume? There are probably other examples but you can hopefully see that the situation or the combination of changes you make, can be as important as the substance you choose. You can determine this by looking at the Sackur-Tetrode equation for the entropy of an ideal gas. It should be possible to reach a higher final temperature but lower final entropy. An ideal gas, starting from an initial state with pressure and volume P i and V i can be subjected to compression (to a volume V f < V i ) and then some surplus heat can be removed while maintaining the volume at V f. Typically when you transfer heat to a substance, its temperature increases and also the entropy of that substance (or the entropy of the system which is just that substance) increases. I apologize for the title, it had to be very compressed to fit in the space available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
